Cat's out of the box as we peer into quantum world
Physicists get to see particles in two states at once, just like the famous Schrodinger paradox, while chemists listen in on private lines to cells
Many people have heard of Schrodinger's cat. The winners of this year's Nobel Prize in physics have shown deep insights into this famous paradox of quantum mechanics.
In addition to the physics Nobel, this week also saw the announcement of the Nobel Prize in chemistry. Both science prizes went to work focusing on the transfer of information. While the prize-winning physics predictably involved brain-bending quantum mechanics, the chemistry prize focused not on test-tube reactions, but on cellular biology.
The Nobel Prize in physics was jointly awarded to Frenchman Serge Haroche, of the College de France and Ecole Normale Superieure, in Paris, and American David Wineland, of the National Institute of Standards and Technology and the University of Colorado Boulder, "for groundbreaking experimental methods that enable measuring and manipulation of individual quantum systems".
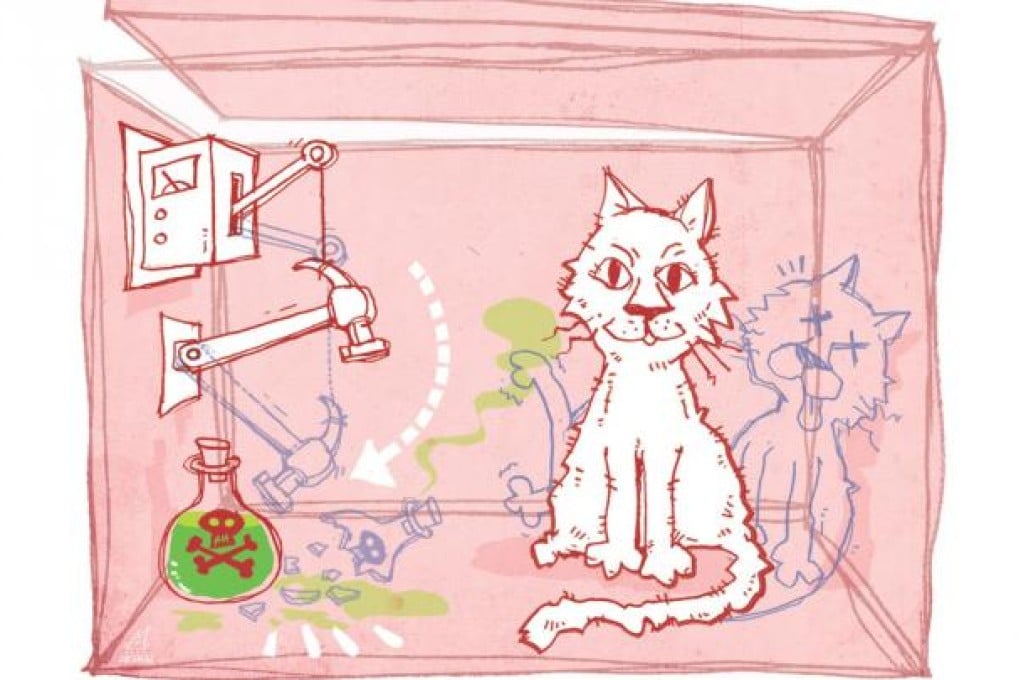
These experiments involve the sort of quantum weirdness described by physicist Erwin Schrodinger in 1935. By extrapolating quantum mechanics to the everyday world, he described a "ridiculous" situation in which a cat was placed in a box with a flask of poison and some radioactive material whose decay could shatter the flask.
According to quantum mechanics, the cat would be both alive and dead at the same time; but of course, if anyone opened the box, they could only find either a live cat or a dead one.
Until recently, it had been thought difficult or impossible to observe individual atoms and sub-atomic particles that might be in two quantum states at the same time: physicists usually studied many particles at once. But sophisticated new techniques, with pioneering work by teams led by Haroche and Wineland, are helping us peer into the quantum world.