Occurrence of multiple-drug-resistant tuberculosis grows
World's second-most fatal disease becomes harder to beat as drug resistance grows and patients fail to follow doctors' orders
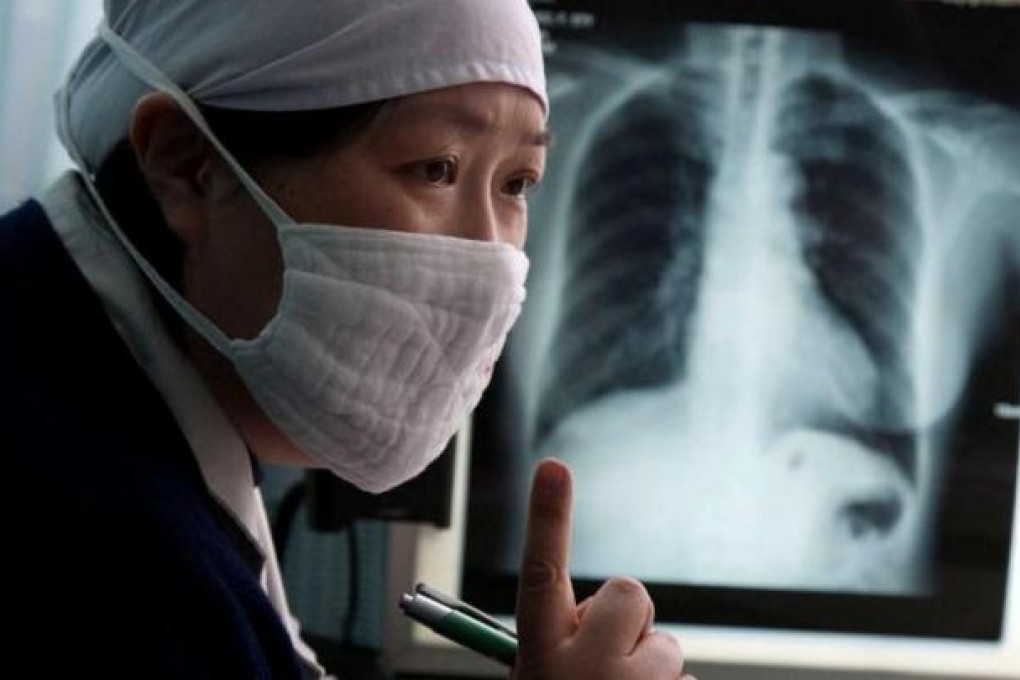
Those fighting tuberculosis (TB) say it is better not to treat the disease than to treat it badly, because doing so just produces more powerful, drug-resistant bacteria, making the world's second-most fatal disease stronger by the day.

Dr Zhao Yanlin, director of the National TB Reference Laboratory, has said that the mainland has a "serious epidemic" of drug-resistant TB, but the situation "could easily be much worse".
Also deputy director of the National Centre for Tuberculosis Control and Prevention at the Chinese Centre for Disease Control and Prevention (CDC), Zhao contributed to a paper published in The New England Journal of Medicine in June about a mainland-wide survey of drug resistant TB.
The survey found that 5.7 per cent of new TB cases and a quarter of previously treated cases were multiple-drug resistant.
Drug-resistant TB emerges when patients fail to follow treatment regimens, which takes six to eight months and use a cocktail of drugs, or stop treatment too soon.