Health officials raise 'outbreak' concerns amid low public confidence in Chinese vaccines
Expert says public vaccination rates have risen very slowly, leaving many people vulnerable to disease
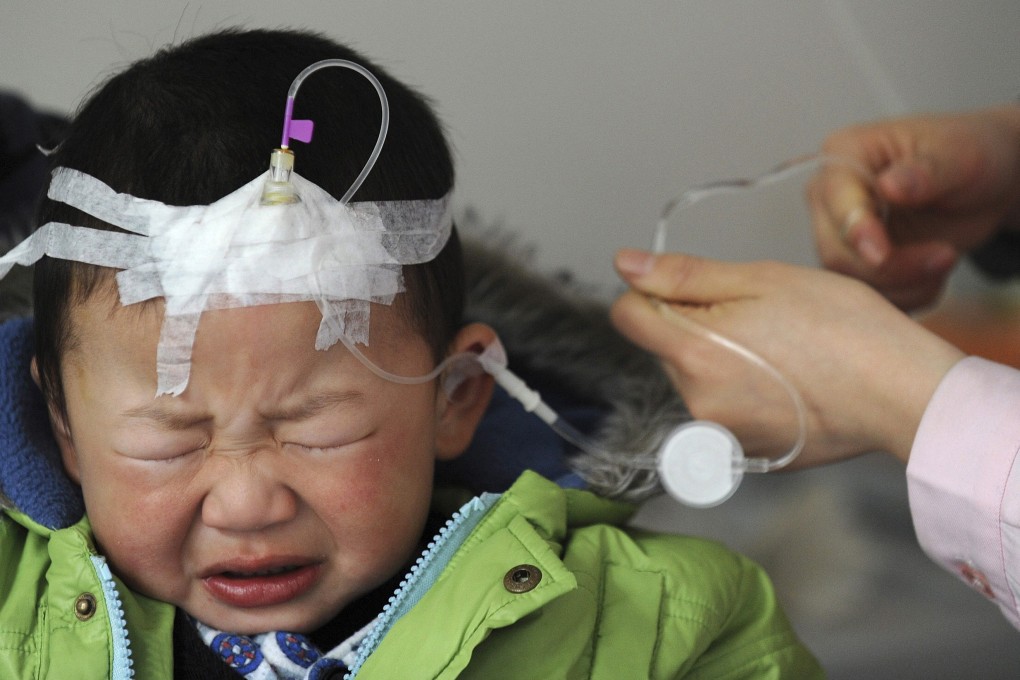
Public confidence in Chinese-made vaccines has recovered painfully slowly in the wake of a hepatitis B death scare, prompting authorities to worry that infectious diseases may gain ground and cause an outbreak.
Hepatitis B shots made by a Shenzhen manufacturer had been suspected of causing the deaths of 17 babies within 24 hours after getting vaccinated in December, but health authorities later ruled out the link and cleared Biokangtai Biological Products of blame.
Li Quanle, a vaccination official with the National Health and Family Planning Commission, told The Beijing News that the vaccination rate had gone up very slowly over the past four months amid lingering doubts over safety.
We aim to restore the public’s confidence in [domestically]-made vaccinations and stop the declining trend
“If the situation continues worsening, there’s a possibility of the outbreak of infectious diseases,” said
A survey by the Chinese Centre for Disease Control and Prevention in January found that the number of hepatitis B vaccinations dropped 30 per cent and other vaccinations had dropped an average of 15 per cent in 10 provinces.
Before that, China’s vaccination ratio since 2006 had been stable at around 98 per cent of the total population, mainly children, required to get inoculations.
The vaccination rate should be more than 90 per cent to ensure that people are shielded from contagious diseases, he said. For some acute contagious diseases like measles, such ratio should be over 95 per cent, according to Li.