Hong Kong doctors will not need to be present all the time in supervising risky beauty procedures
Government sources also say medical practitioners will not have to have specialist qualifications to oversee these procedures
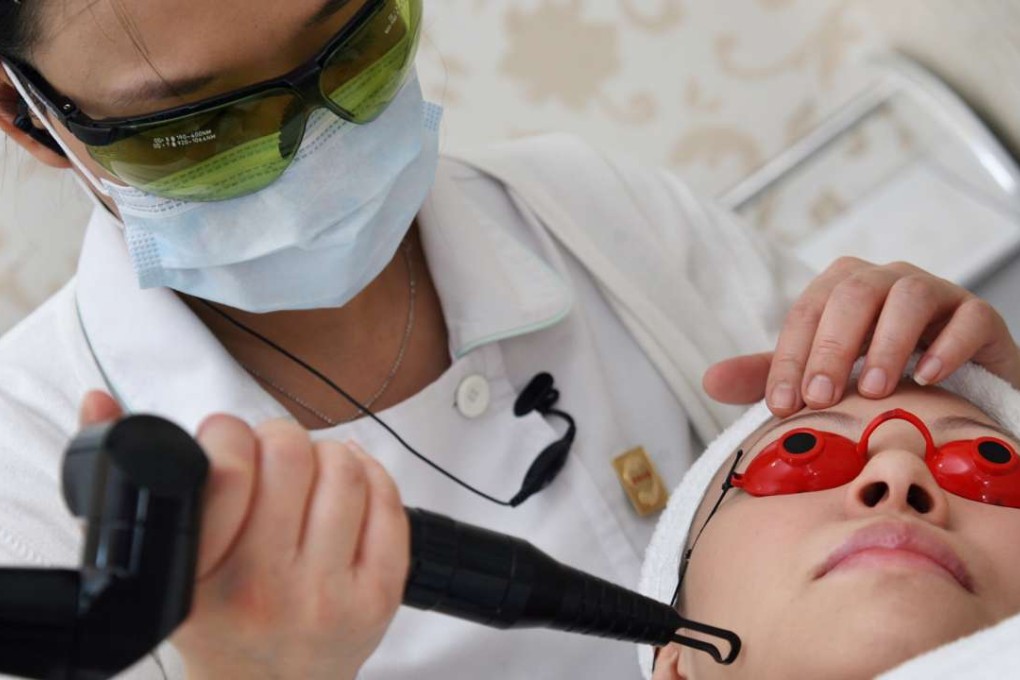
Doctors will not be required to be present throughout beauty procedures involving the use of risky medical devices or have specialist qualifications, according to a government proposal.
The clarification came from government sources ahead of a Legislative Council meeting on the regulation of medical devices on the coming Monday.
This came after the beauty sector expressed strong opposition to the strict regulation of devices in common use.
Under the government’s proposal, 15 types of devices that are most commonly used by non- medical practitioners for non-medical purposes, such as beauty treatment, will be regulated according to two levels of risk.
Sources said beauticians, who have completed government-recognised training, could either use the devices under the supervision of a registered doctor for riskier procedures or perform less risky ones alone.