Why Chinese biotech companies are not for faint-hearted investors
- CK Life Sciences is up 97 per cent since announcing favourable clinical trial results for a skin cancer drug four weeks ago
- But some analysts prefer proven big players with approved drugs and large marketing teams

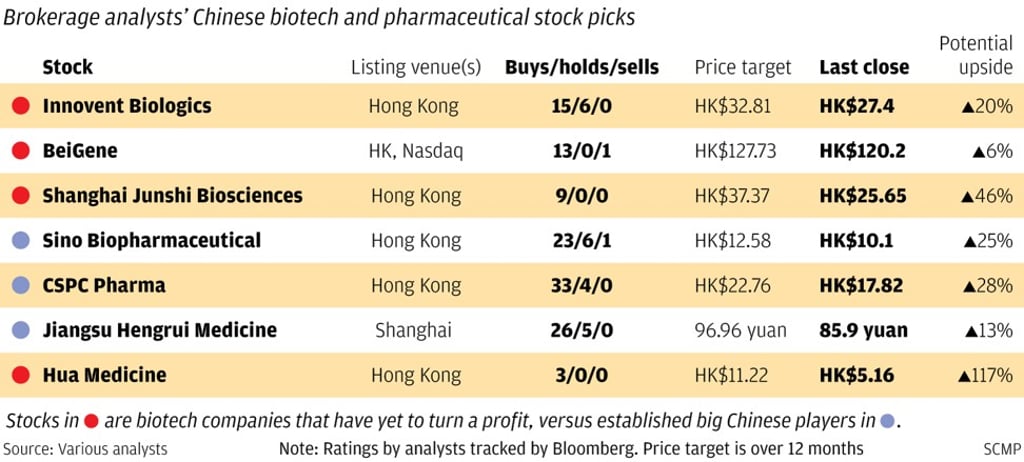
Chinese biotechnology companies that show promise but have yet to turn a profit are not for faint-hearted investors. Their share prices can be extremely volatile.
Take Shanghai-based diabetes drug developer Hua Medicine. Its share price plunged by 40 per cent over three days recently after interim clinical trial results for its diabetes drug disappointed investors. Then it shot up 30 per cent over the following two days after reassurances by its executives.
Companies like Hua Medicine are searching for the next breakthrough treatments for serious, sometimes fatal diseases.
While few treatments ever make it through the regulatory approval process, those that do can produce huge rewards.
Alan Auerbach, for example, became an overnight billionaire five years ago when his US-listed Puma Biotechnology’s breast cancer drug showed success in phase three clinical trials.
But is it worth the risk to invest in such unproven but innovative drug companies? And how can investors without a deep science background understand results of clinical trials to know whether to wait, bail or jump into a stock?