Li Ka-shing-backed Israeli biotech start-up’s test to distinguish bacterial and viral infections wins US FDA approval
- The US Food and Drug Administration approves the first test that can accurately identify the nature of infection in 15 minutes
- Doctors say MeMed’s test will help cut unnecessary prescriptions of antibiotics

The first-of-its kind test – MeMed BV – decodes the immune response in 15 minutes to accurately identify the nature of infection and has been cleared for use for both children and adults. Previously, bacterial and viral infections were often clinically indistinguishable leading to overprescription of antibiotics, which are ineffective against diseases caused by viruses.
Overuse of antibiotics drives the emergence of antimicrobial resistance, a process whereby bacteria and viruses change over time and become resistant to medicines, making them harder to treat and increasing the risk of the severity of the illness and its ability to spread.
“[Antimicrobial resistance] is arguably today one of the biggest health care challenges of our time and it has exacerbated because of Covid,” said Eran Eden, co-founder of MeMed, adding that it will continue to grow even after the pandemic subsides unless “we do something very dramatic to fight it”.
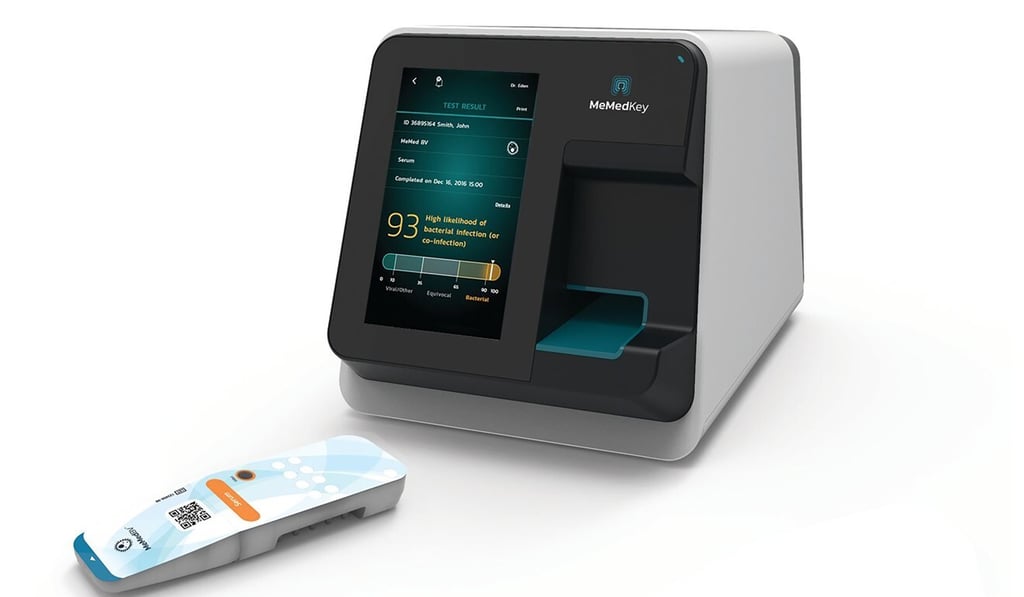
Launched in 2009 by Eden and Kfir Oved, MeMed has since received investments totalling US$100 million from venture capital firms. Horizon Ventures, MeMed’s largest shareholder, bought its stake in 2013.
Eden, who is also the CEO, said he was confident MeMed will launch an initial public offering soon. “That’s definitely one of the things that we’re considering seriously with our partners, [including] Horizons,” he said, without elaborating.