Coronavirus: India reviews AstraZeneca vaccine side effects; Thailand’s Koh Samui eyes October reopening
- New Delhi decided to conduct the review after several countries suspended their roll-outs of the AstraZeneca jab over blood clot fears
- Elsewhere, the Philippines has detected its first case of the Brazil variant, while the resort island of Koh Samui plans to reopen to vaccinated from October 1
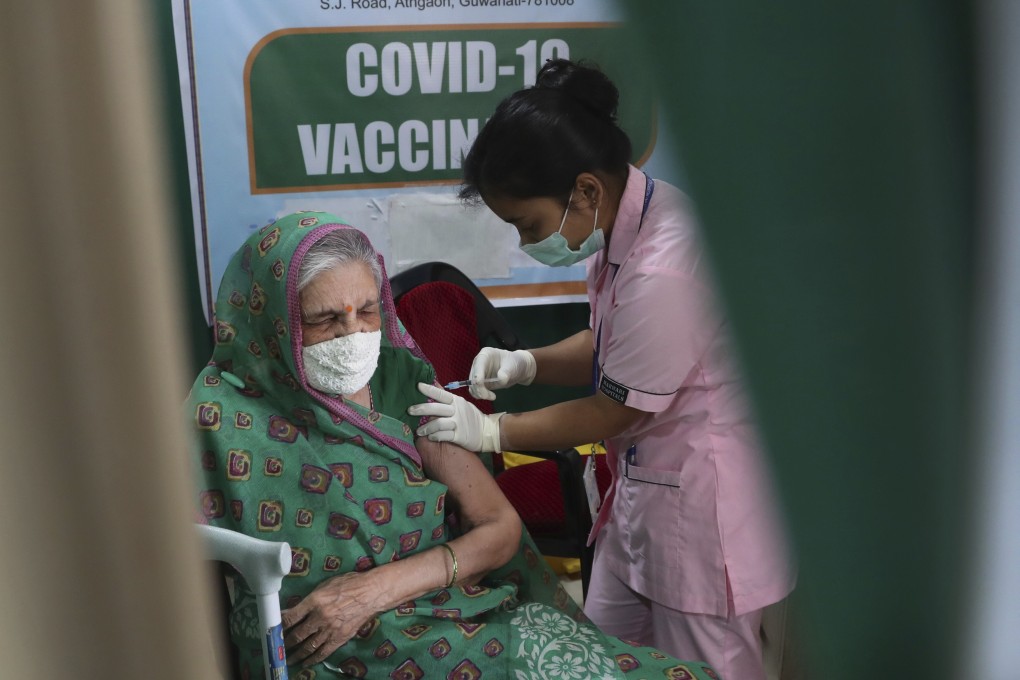
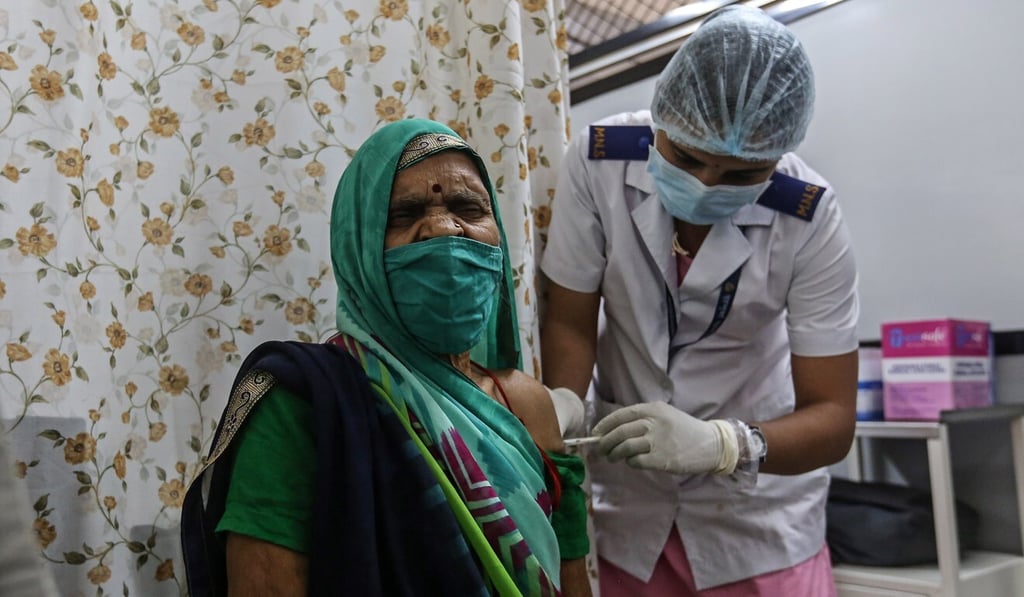
India will carry out a deeper review of post-vaccination side effects from the AstraZeneca coronavirus shot next week although no cases of blood clots have been reported so far, an official said on Saturday.
“We are looking at all the adverse events, particularly serious adverse events like deaths and hospitalisation. We will come back if we find anything of concern,” said NK Arora, a member of India’s national task force on Covid-19.
India has given at least 28 million shots in its vast vaccination programme, most of them AstraZeneca’s which are produced at the Serum Institute of India.
New Delhi has also gifted and allowed exports of millions of these jabs to around 70 countries over the last few weeks as a part of its vaccine diplomacy.
Arora said there was “no immediate issue of concern as number of adverse events (in India) is very, very low. We are relooking at (adverse events that were reported) to see if there was any issue of blood clotting”.