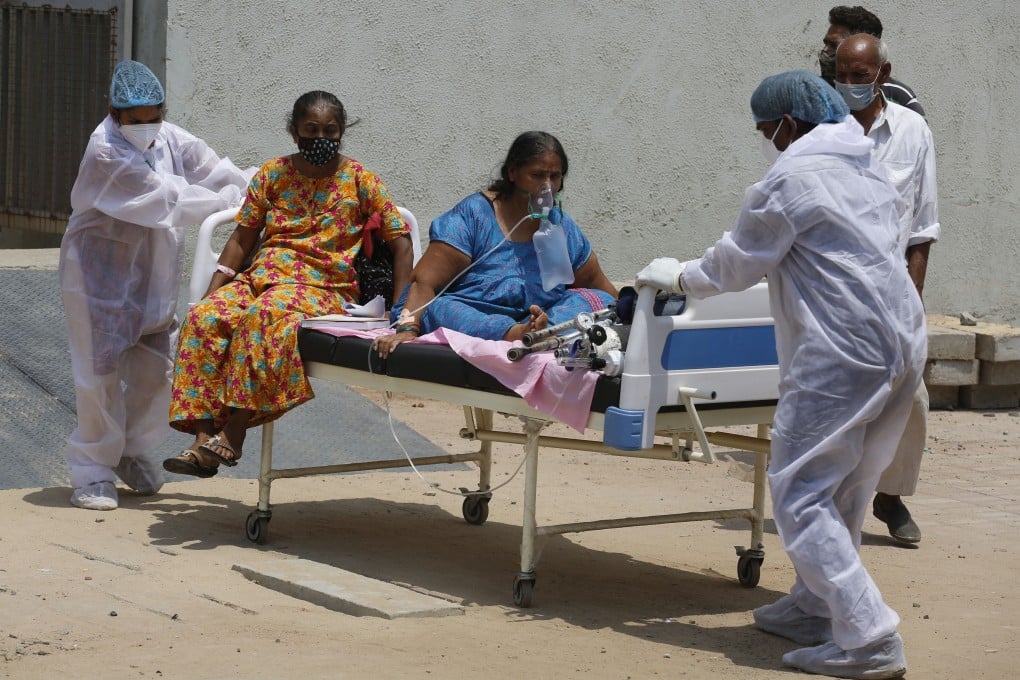
Coronavirus: India to fast track vaccine approvals; Indonesia satisfied with Sinovac
- India, which has overtaken Brazil as the second hardest-hit country, will grant emergency use approval to vaccines authorised in other countries
- Indonesia said local trials found the Chinese-made vaccine 65 per cent effective, while Philippine leader Rodrigo Duterte will ‘waive’ his vaccine opportunity
The move, which will drop the need for companies to do small, local safety trials for their vaccines before seeking emergency approval, follows the world’s biggest surge in cases in the country this month.
Vaccines authorised by the World Health Organization or authorities in the United States, Europe, Britain and Japan “may be granted emergency use approval in India, mandating the requirement of post-approval parallel bridging clinical trial”, the health ministry said.
“The first 100 beneficiaries of such foreign vaccines shall be assessed for seven days for safety outcomes before they are rolled out.”
India, the world’s biggest maker of vaccines, has administered more than 106 million doses of Covid-19 shots, but many regions are now running short of supplies as inoculations expand due to surging cases. It has sold more than 54.6 million vaccine doses abroad and gifted more than 10 million to partner countries.
It is currently using the AstraZeneca shot and a home-grown vaccine developed by Bharat Biotech for its own immunisation drive, and this week approved Russia’s Sputnik V shot for emergency use.