Advertisement
How joining vaccine initiative Covax could be shot in the arm for China
- Adding its weight to the WHO-led alliance is also a way of safeguarding China’s own access to vaccines
- Engaging with global bodies and standards could aid acceptance of vaccines produced in China after past scandals
4-MIN READ4-MIN
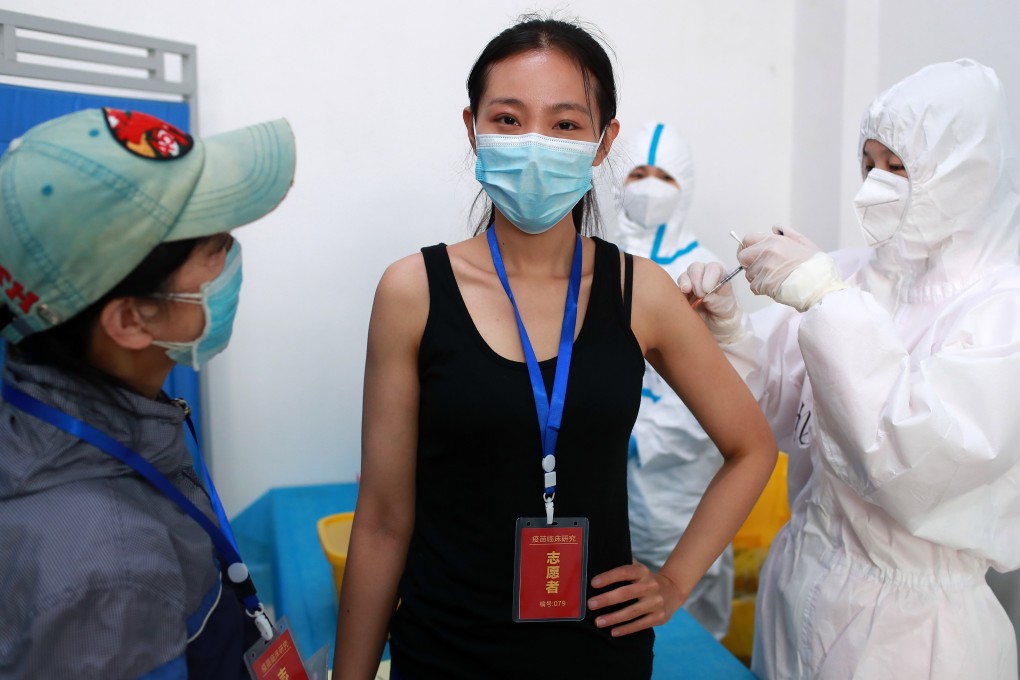
China’s decision to join Covax was seen as a boost to the global scheme to ensure fair access to Covid-19 vaccines for poorer countries, but membership could benefit itself as well as any nations receiving its doses.
Joining the initiative, after having initially opted out, will give China access to vaccines not previously available in its domestic market and could help its scandal-plagued vaccine industry gain the international recognition it has lacked, experts said.
Four Chinese candidates are in the final stage of human trials in the global race for a Covid-19 vaccine, while seven other Chinese candidates are in different stages of trials. Production facilities have been built in preparation, with capacity expected to reach 610 million doses by end of this year and 1 billion next year – expected to meet China’s own inoculation needs if those vaccines have been approved.
Advertisement
Nonetheless, the Chinese government has agreed to buy vaccines for 15 million people through Covax, and has boosted the initiative’s resources by pledging to deliver at least 2 billion doses of China-made vaccines, if approved, by the end of next year to be available to its 172 member countries.
02:40
If China’s coronavirus vaccines work, which countries will get them and for how much?
Insurance policy
Advertisement
Advertisement
Select Voice
Select Speed
1.00x