Advertisement
What happens next, as coronavirus vaccine trials move to a new phase?
- Teams from China, Britain, the US and Germany have shown their potential products are safe but there’s still a long way to go
- Broad testing across a diverse range – including elderly and less healthy subjects – is crucial to assessing how a vaccine might work on a population
4-MIN READ4-MIN
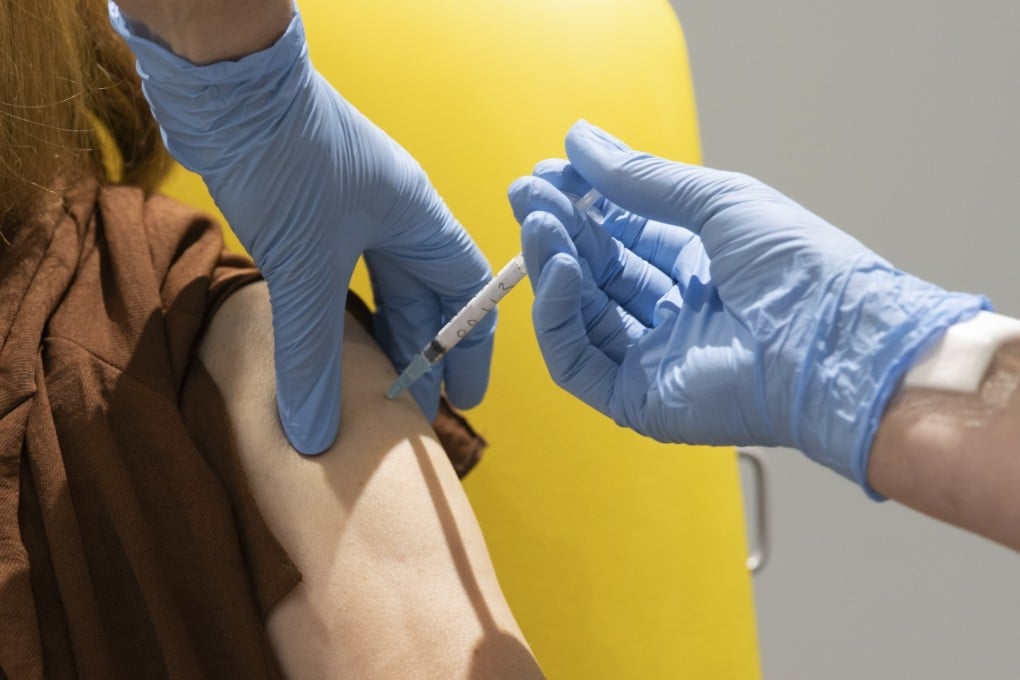
The race for a Covid-19 vaccine has taken on critical importance as the disease continues to charge through the global population, with more than a million new infections every week and the death toll climbing.
The vaccine has been seen as a way out of the crisis and new results from some of the teams leading development are showing early promising signs. But the real test lies ahead in the final-phase trials, experts say.
The new data out on Monday includes results from a vaccine candidate produced by a team at Oxford University, working in partnership with British firm AstraZeneca.
Advertisement
The candidate was found to be safe and induced strong immune responses in combined phase-one and phase-two trials, published in The Lancet scientific journal.
A candidate from Chinese biopharmaceutical company CanSino Biologics, produced in collaboration with the Chinese military, was also found to be safe and able to produce an immune response in most candidates, according to results from phase-two trials, also published in The Lancet on Monday.
Advertisement
Advertisement
Select Voice
Select Speed
1.00x