Advertisement
Chinese vaccine firms to rush 110 million doses to Covax now, half a billion by mid-2022
- Sinovac and Sinopharm sign advance purchase agreements to provide more doses and diversify the Covax portfolio to help developing nations beat the pandemic
- Gavi, the Vaccine Alliance has the option to extend its purchase of Chinese vaccines, alleviating a shortfall created by the virus surge in India
3-MIN READ3-MIN
53
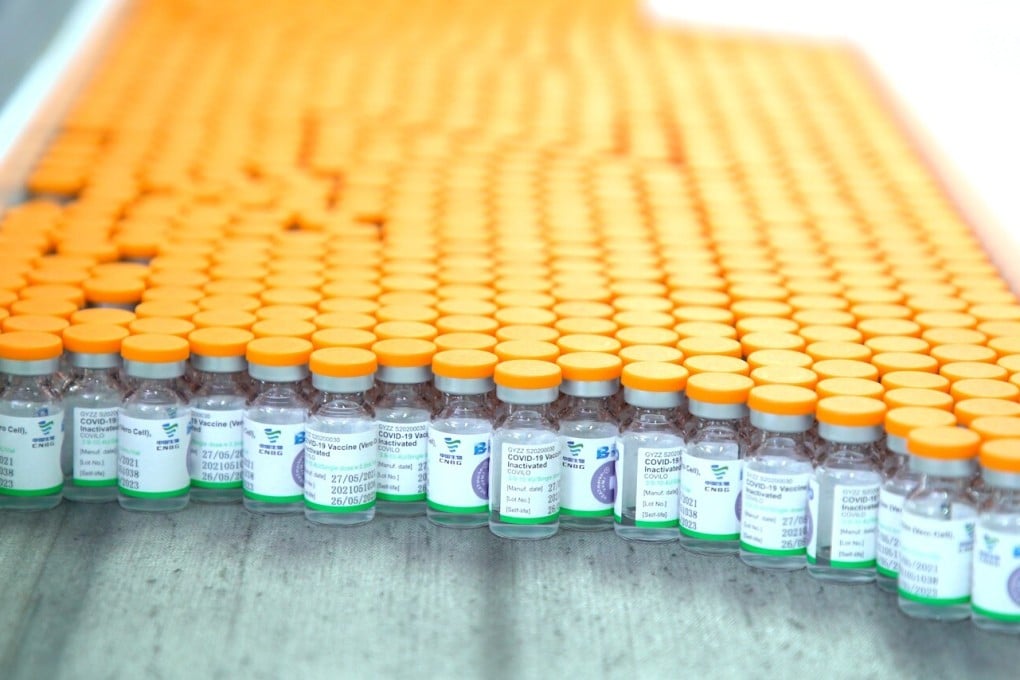
A global sharing scheme has contracted two Chinese manufacturers to immediately supply 110 million coronavirus vaccine doses to poorer countries as part of an advance purchase agreement.
The agreements with Sinopharm and Sinovac – which have been validated by the World Health Organization for emergency use – come as the Delta variant, a more transmissible strain expected to become dominant globally, poses a rising risk to health systems.
The purchase agreements are expected to make up the shortfall of doses seen by the Covax Facility, a global sharing scheme aiming to provide equitable access to Covid-19 vaccines.
Advertisement
Gavi, the Vaccine Alliance, a public-private partnership which buys vaccines for Covax, said in a statement on Monday that 60 million doses from Sinopharm would be made available between this month and October and Sinovac would provide 50 million doses of its vaccines by the end of September.
Gavi has the option to buy a further 60 million doses in the fourth quarter this year and 50 million more doses in the first half of next year from Sinopharm. In the agreement with Sinovac, Gavi can buy a further 150 million doses in the fourth quarter of 2021 and 180 million more doses in the first half of 2022.
Advertisement
Advertisement
Select Voice
Select Speed
1.00x