Advertisement
Coronavirus: China to start vaccinating children as young as 3 amid new outbreaks
- Governments in at least five provinces have said vaccinations will be required for those aged from three to 11
- It comes as parts of the country take measures to try to stop the spread of cases as authorities maintain zero-tolerance policy
3-MIN READ3-MIN
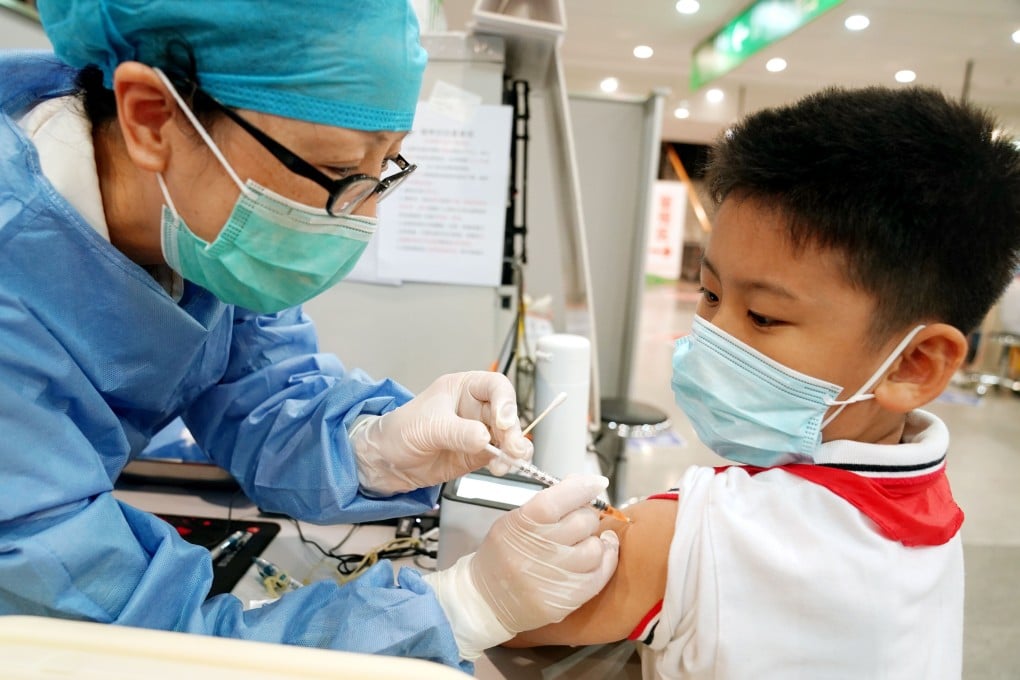
Children as young as three will start receiving Covid-19 vaccines in China, where 76 per cent of the population has been fully vaccinated and authorities are maintaining a zero-tolerance policy towards outbreaks.
Local city and provincial-level governments in at least five provinces issued notices in recent days announcing that children aged from three to 11 will be required to get vaccinated.
The expansion of the vaccination campaign comes as parts of China take new clampdown measures to try to stamp out small outbreaks. Gansu, a northwestern province heavily dependent on tourism, closed all tourist sites on Monday after finding new Covid-19 cases. Residents in parts of Inner Mongolia have been ordered to stay indoors due to an outbreak there.
Advertisement
The National Health Commission reported 35 new cases of local transmission had been detected over the past 24 hours, four of them in Gansu. Another 19 cases were found in the Inner Mongolia region, with others scattered around the country.
China has employed lockdowns, quarantines and compulsory testing for the virus throughout the pandemic and has largely stamped out cases of local infection while fully vaccinating 1.07 billion people in its population of 1.4 billion.
Advertisement
In particular, the government is concerned about the spread of the more contagious Delta variant by travellers and about having a largely vaccinated public ahead of the Beijing Winter Olympics in February. Overseas spectators have already been banned from the Games, and participants will have to stay in a bubble separating them from people outside.
Advertisement
Select Voice
Select Speed
1.00x