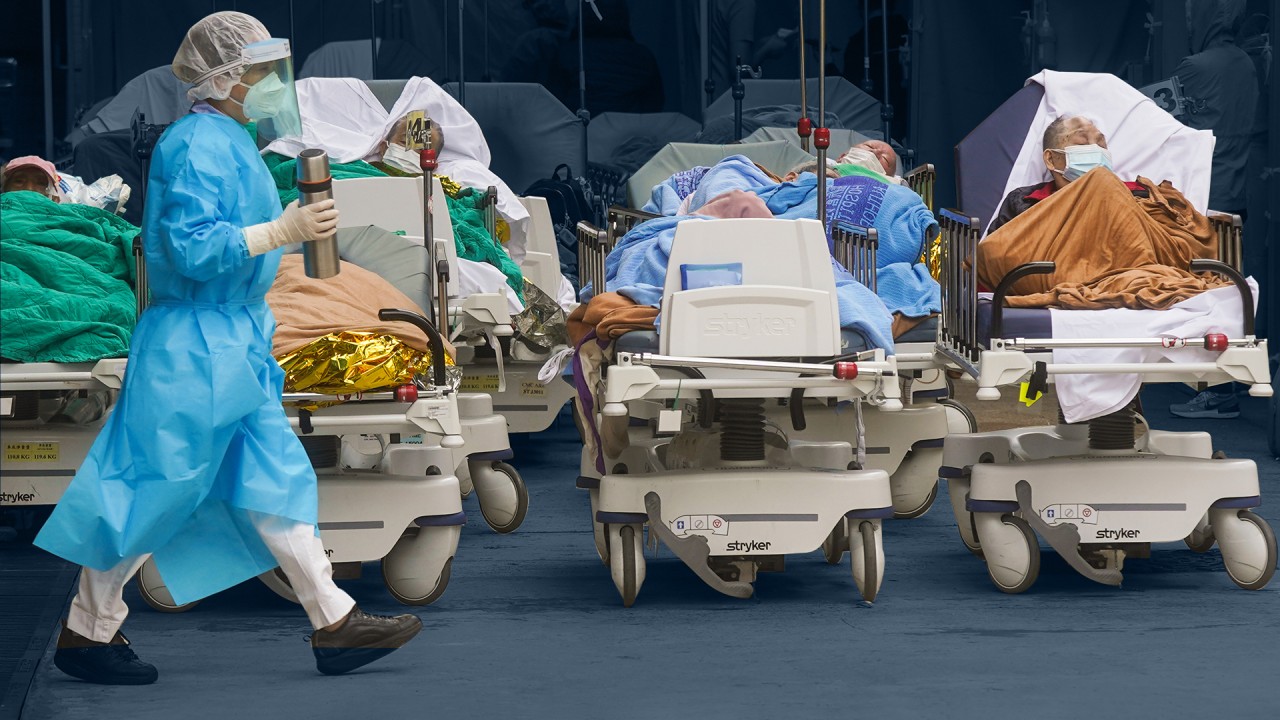
Hong Kong in talks with mainland Chinese firm over Covid-19 treatment
- Brii Bioscinences confirms it is in talks about supplying a cocktail of drugs to the city, which is battling a wave of cases
- The treatment has been approved for use in mainland China and recommended by leading respiratory diseases expert Zhong Nanshan
Top officials are handling Hong Kong’s Covid crisis from a Shenzhen villa
Zhong also urged the city to consider other treatments such as the US-made Paxlovid and traditional Chinese medicine, adding that he had spoken to businesses on the mainland about offering greater support to the city.
Brii’s treatment, a combination of monoclonal neutralising antibodies, was approved in Hong Kong for clinical trials last November but is yet to be approved for registration.
In December mainland drug regulators approved the therapy for adults who are at high risk of developing severe Covid-19 and conditional approval for patients aged 12 to 17. Several provinces had previously allowed its use on compassionate grounds.
In early November a statement from the company said the treatment had been given to more than 700 patients for free, the oldest of whom was 92.
The treatment, administered by infusion, was co-developed with Tsinghua University in Beijing and the Third People’s Hospital in Shenzhen.
The Tsinghua team, led by Professor Zhang Linqu, screened B cells, a type of white cell that produces antibody molecules, from infected patients in the hospital in Shenzhen and selected ones which bind with part of the coronavirus’s spike protein – an indicator they could neutralise viruses.
The team found a total of 206 antibodies, had their genomes sequenced to find the best candidate and reconstructed them in a lab.
The drug showed an 80 per cent reduction in hospitalisations and deaths compared with placebos in non-hospitalised patients.
In December, the company said in a statement that lab studies showed the combination therapy holds up well against Omicron and other variants of concern, including Delta.
Hong Kong mum ‘cries, begs’ after hospital separates her from baby with Covid-19
The therapy is under review by the Food and Drug Administration for emergency use in the United States, one of its clinical trial sites. Other trial sites were in Argentina, Brazil, Mexico, South Africa and the Philippines.