
Chinese execs took firm’s Covid-19 vaccine pre-trials ‘to lead by example’
- China National Biotec Group tried its experimental vaccines on 180 executives before human trials were approved
- ‘My arm went blue from drawing blood dozens of times,’ the company’s vice-president Zhang Yuntao says
The volunteers included Sinopharm chairman Liu Jingzhen, along with CNBG’s chairman Yang Xiaoming and vice-president Zhang Yuntao, who took shots in March.
“We are very confident in our own vaccines,” Zhang said. “We are in charge of the vaccine development and we need to lead by example, so we tested the vaccine on our own bodies.”
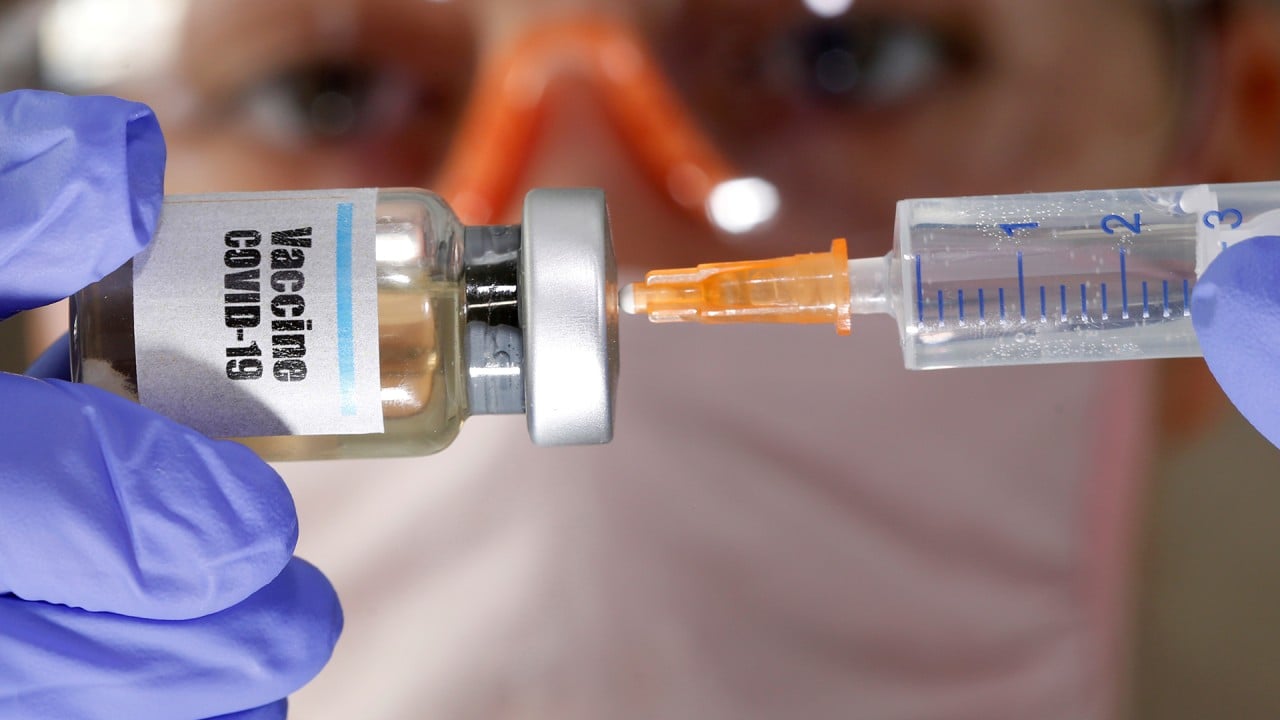
06:17
‘Robust immune responses’ found in Covid-19 vaccine clinical trials point to 2021 release
Testing drugs on one’s employees is deemed unethical under modern pharmaceutical protocols, but sometimes in China it is considered a noble sacrifice.
Chinese media have recounted how Nobel laureate Tu Youyou tested an anti-malaria drug on herself in 1972 and Gu Fangzhou, a Chinese virologist who developed a domestic oral polio vaccine, tested it on his baby son.
Zhang said the CNBG and Sinopharm executives were not part of clinical trials, which are randomised and controlled, but had been monitoring antibody levels to try to gauge the vaccines’ effectiveness. The phase 1 and 2 trials for CNBG’s two Covid-19 vaccines were conducted in China from April to June, and the data was published in the journal Cell and the Journal of the American Medical Association.
Do Chinese Covid-19 vaccines work, and which countries will get them?
“We couldn’t test directly the protectiveness of the vaccines, but we can compare the antibody levels with those of the recovered Covid-19 patients,” Zhang said. “The antibody level and duration [of high antibody level] are even higher. My arm went blue from drawing blood dozens of times.”
CNBG is testing two vaccines in various countries for safety and protectiveness data, with which the company can apply for regulatory approval. Although protectiveness data was not yet complete, the safety data was very satisfactory, Zhang said.
None of the announced vaccine candidates selected by Warp Speed used actual viruses. Some used mRNA (messenger ribonucleic acid), which tells cells how to make proteins. An mRNA vaccine is designed to mock an attack by the protein spikes with which the coronavirus invades human cells, and trigger an immune response to any future coronavirus-infected cells carrying the same spikes.
Zhang said CNBG had considered mRNA vaccines before choosing the more traditional inactivated option.
“There has been no approval of mRNA vaccines for humans before and its safety needs long-term monitoring,” Zhang said. “Covid-19 vaccines are not simply about technological innovation. It will be used on healthy people of all age groups, so safety comes first.”
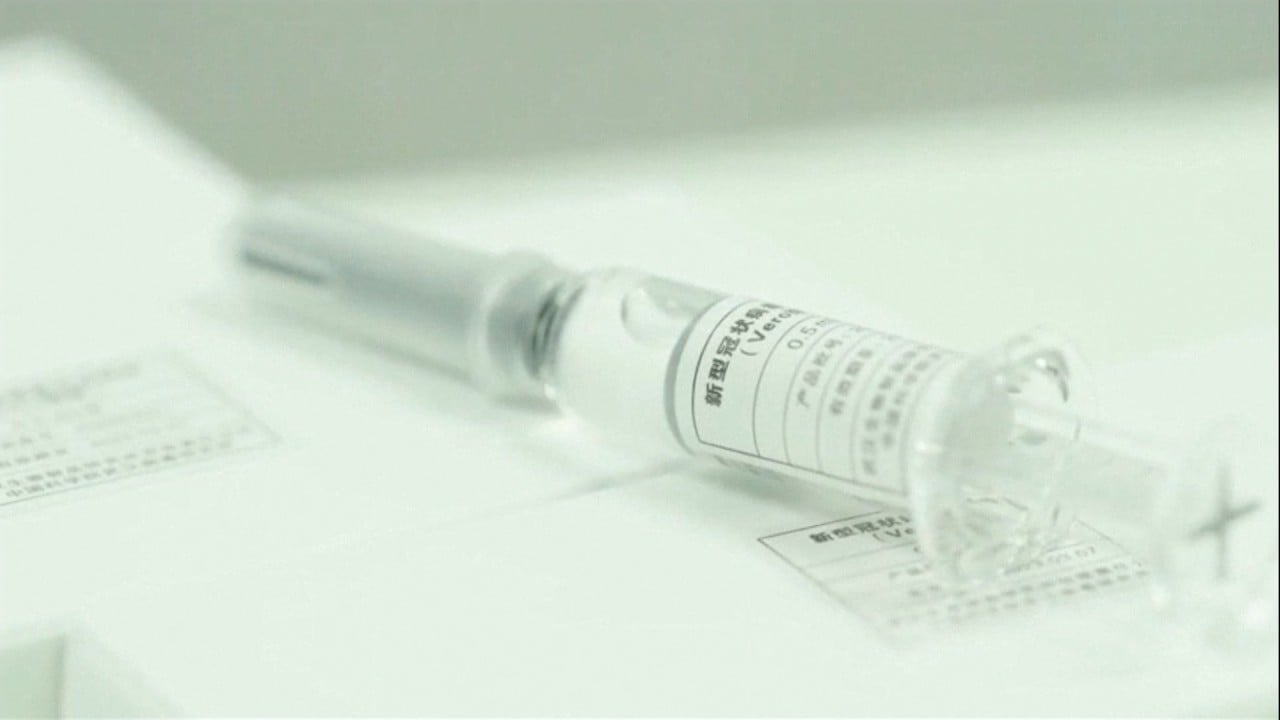
01:39
China prepares for coronavirus vaccine mass production though clinical trials are not yet complete
“Having the trial in multiple places is to test the vaccine among different [racial and ethnic] groups,” he said. “More continuous monitoring [of safety data] is required before such vaccines get full approval.”
Even longer monitoring will be needed after the approval, if received. Administering a vaccine to a large population can expose antibody-dependent enhancement (ADE): antibody protection that has the potential to amplify the infection or trigger harmful immune responses.
“Clinical trials will not be the end of the vaccine development,” Zhang said. “It needs continuous monitoring.”

