Using only two fingers, Hong Kong girl types 22-page proposal calling for life-saving muscle drug
Josy Chow Pui-shan, 23, wants government to look into new but costly medication for spinal muscular atrophy patients
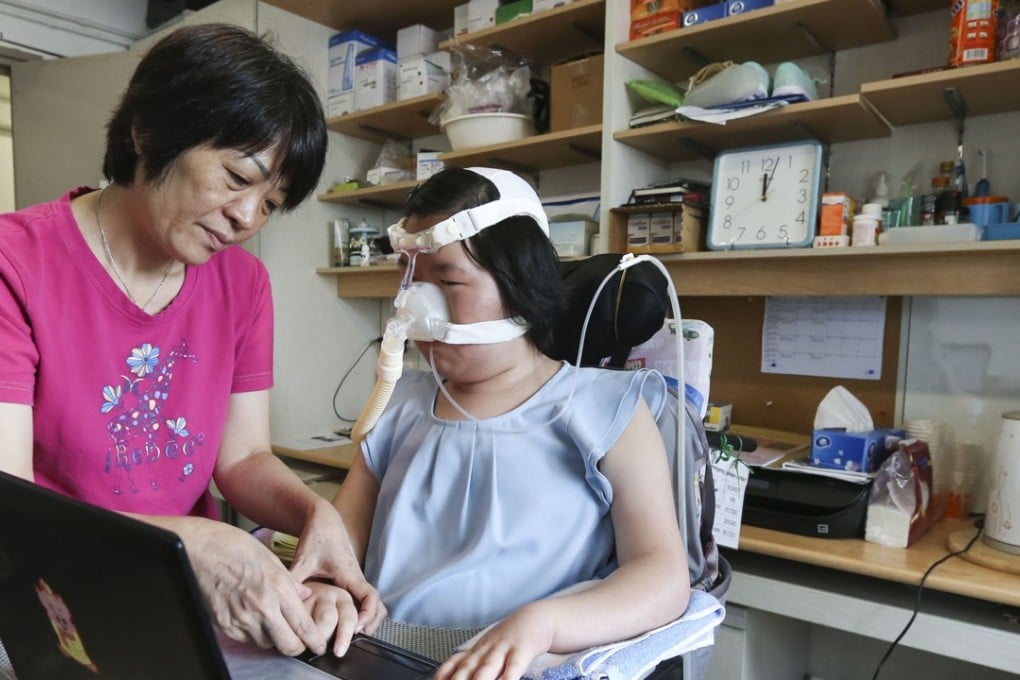
Using just two fingers, a 23-year-old Hong Kong woman who is almost completely paralysed by a rare disease has written a proposal calling for patients to be treated with a new drug that could be life-saving.
Josy Chow Pui-shan, who was born with spinal muscular atrophy (SMA), a genetic disorder that gradually destroys physical abilities such as walking and breathing, urged the government to introduce nusinersen, marketed as Spinraza, the first approved drug for SMA by the US Food and Drug Administration.
She will submit the proposal in a petition on Sunday staged by SMA patients and families outside the office of the chief executive in Admiralty.
Disabled students battle against the odds in Hong Kong’s pre-university exams
The therapy, approved in December last year, was deemed as a lifesaver for the incurable disease. But it has not been registered in Hong Kong yet.
Typing with her right thumb and right index finger on a special mouse pad, Chow, an English and linguistics student at the University of Hong Kong, could only form four to five words a minute on her laptop, and needed to rest for five to 10 minutes between lines.
“It is really tiring to write with two fingers only ... but I don’t want other children [in a similar condition] to experience the hardship I have had growing up,” Chow said.

To cope with her breathing difficulties, she wears a face mask for ventilation and has a plastic suction tube in her mouth to clear away mucus in her throat.