ExplainerCovid-19 vaccine: how soon will the shots be available in Hong Kong and mainland, and what are the possible side effects?
- A timetable for vaccination is yet to be fixed, but delivery of the first batch of 1 million jabs from Sinovac – originally expected in January – could be delayed
- Chinese experts are developing mRNA, inactivated and nucleic acid-based vaccines, while more than 1.5 million people have already received the shots
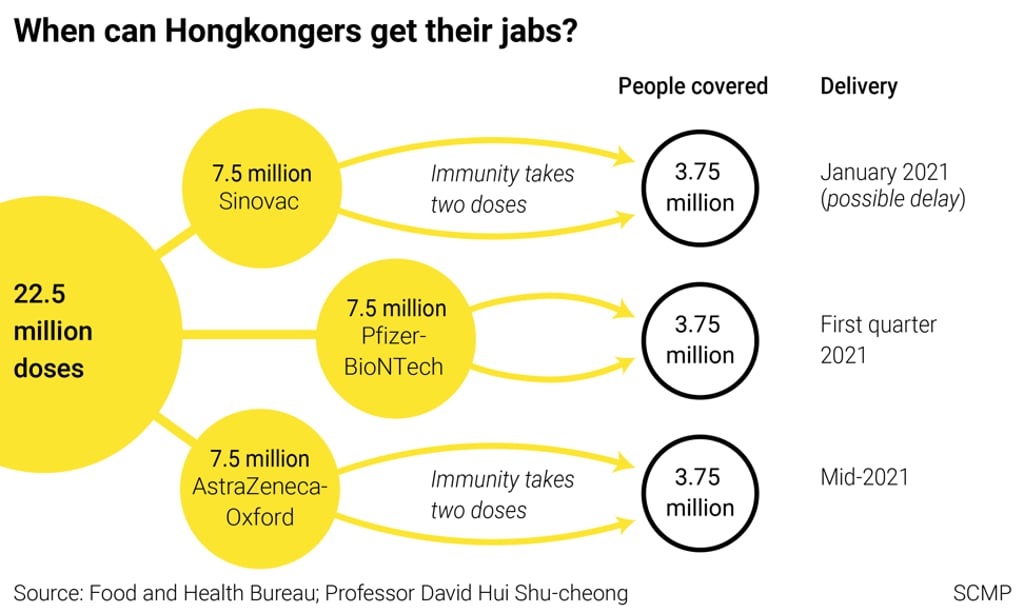
What Covid-19 vaccines will be available in Hong Kong?
Hong Kong has so far struck deals to purchase 22.5 million doses of Covid-19 vaccines, with 7.5 million doses each coming from three suppliers: Beijing-based Sinovac Biotech; Fosun Pharma which offered the vaccine co-developed by Germany’s BioNTech and US-based Pfizer; and British-Swedish firm AstraZeneca.
Scientists from the University of Hong Kong, together with researchers in mainland China, are also developing a nasal spray vaccine against Covid-19, the first such medication approved for testing on humans.
When can I be vaccinated?
An exact timetable for the vaccination scheme has not yet been fixed. While the first batch of 1 million jabs from Sinovac were originally expected to arrive in January, that could be delayed, as the mainland drug maker had postponed the announcement of its phase 3 clinical data, which was first expected on December 23.