Hong Kong researchers say hepatitis C drug can prevent coronavirus from replicating, might help in Covid-19 treatment
- Antiviral drug simeprevir works against coronavirus, lab tests show
- No longer used for hepatitis C, drug can be ‘repurposed’ quickly and at lower cost for Covid-19
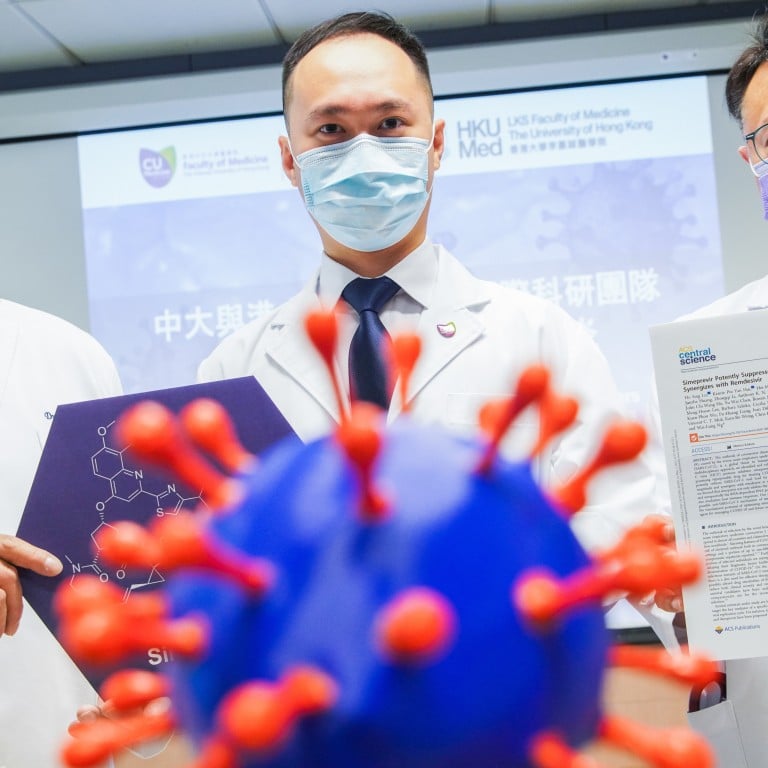
A study led by scientists in Hong Kong has found that a hepatitis C drug may improve the treatment options for patients with Covid-19.
Laboratory tests showed that the drug, simeprevir, is able to prevent the Sars-CoV-2 coronavirus that causes Covid-19 from replicating.
The team of 30 scientists from local and overseas laboratories, led by two professors from Chinese University (CUHK) and the University of Hong Kong (HKU) has begun animal trials.
They said the drug had already been cleared by the United States’ Food and Drug Administration (FDA) and if all went well, it could be used to treat Covid-19 patients as early as next year.
“Repurposing an existing FDA-approved drug can shorten the time frame for clinical trials,” said Billy Ng Wai-lung, assistant professor of pharmacy at CUHK.
“We believe the cost of producing the drug will not be too high because it has passed the R&D stage. The benefit of using an existing drug is the ability to skip the most expensive part of the process.”
Simeprevir was used to treat hepatitis C until 2018, when it was taken off the market after newer drugs emerged for the liver infection caused mainly by a blood-borne virus.
The researchers found a new use for the drug by observing its effect on two viral proteins critical to replicating the virus – the main protease (Mpro) and RNA polymerase.
Simeprevir targets two viral proteins at the same time, so it is quite difficult for the virus to simultaneously develop two mutations on two different proteins
Ng said Mpro acts like a pair of molecular scissors that cuts a chain of proteins at specific sites, to enable the virus to mature and make new copies. RNA polymerase, meanwhile, works like a photocopier that reproduces the genetic material of the virus, allowing it to replicate.
Simeprevir is believed to be the only antiviral drug that can target more than one protein in the coronavirus, remaining effective even if one of the proteins becomes mutated.
Currently, Covid-19 patients with mild symptoms are given a cocktail of antiviral drugs, while those who are seriously ill are prescribed the drug remdesivir.
Expert in vaccine-mixing research to study case of man who got three doses involving Sinovac, BioNTech
Ng pointed out that remdesivir targets RNA polymerase only, meaning viral mutants resistant to the drug could still emerge.
“To develop remdesivir resistance is easier, because you just need one mutation to make the drug ineffective,” he said. “But simeprevir targets two viral proteins at the same time, so it is quite difficult for the virus to simultaneously develop two mutations on two different proteins.”
The team found that using simeprevir together with remdesivir increased the anti-viral effect more than 100-fold compared with using remdesivir alone, and combining both drugs could also help lower treatment costs and reduce side effects.
Their findings were published last month in ACS Central Science, a leading international chemistry journal.
The ongoing animal trials are expected to run between six months and a year.
Michael Chan Chi-wai, associate professor of public health at HKU, said that during this stage, the team would test whether simeprevir was effective against mutated strains of the coronavirus in animals.
Ng said that the research could also help prepare for future pandemics.
“We’ve already seen the emergence of severe acute respiratory syndrome (Sars) and Middle Eastern respiratory syndrome (Mers), which were both caused by coronaviruses. Having the data on the drug readily available will be helpful, as coronaviruses have been identified as pathogens with pandemic potential,” he said.

