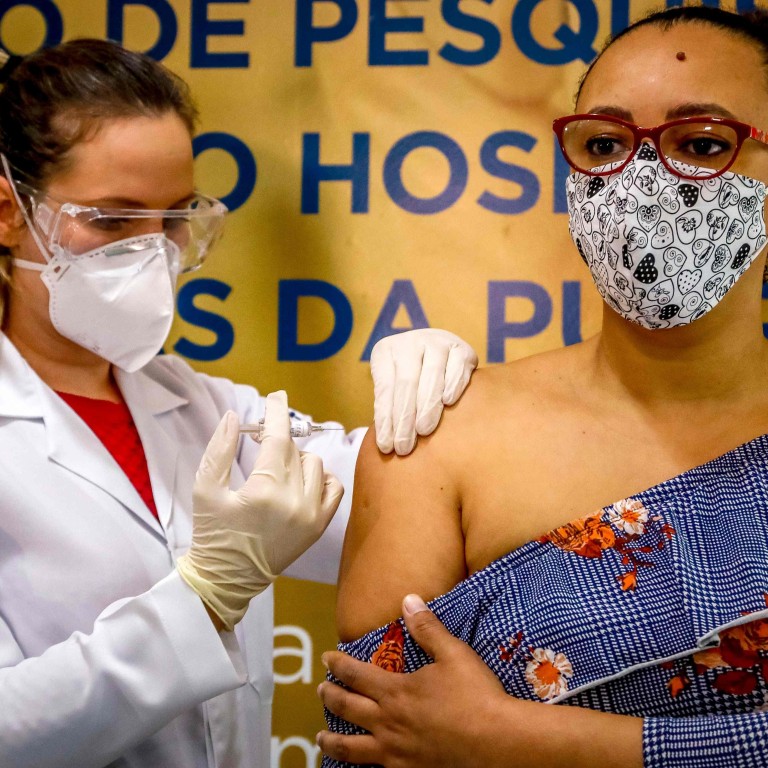
Brazil allows trials of Chinese CoronaVac vaccine to resume
- Far-right President Jair Bolsonaro, who has criticised the vaccine ‘from that other country’, had claimed the suspension of trials as a victory
- However, public health officials said a volunteer recipient’s death, which police are investigating as a suicide, had no connection with the vaccine.
The regulatory agency, Anvisa, said it had now received more details on the nature of the “adverse incident” that led it to halt trials of Sinovac Biotech’s CoronaVac vaccine, and had “sufficient information to allow vaccination to resume.”
Brazil spat over China vaccine is more about politics than health policy
Far-right President Jair Bolsonaro, who has criticised the vaccine “from that other country,” had claimed the suspension as a victory. The president, known as “Trump of the Tropics” for his populist platform, has said Brazilians were being used as guinea pigs for CoronaVac and questioned its safety.
However, public health officials said the “adverse incident” that led to the suspension – a volunteer recipient’s death, which police are investigating as a suicide – had no connection with the vaccine.

“The Brazilian government fears a Chinese retaliation and [Bolsonaro’s] discourse is purely ideological, it is not tied to reality,” Caramuru de Paiva said.
CoronaVac has been caught up in a messy political battle in Brazil, where its most visible backer has been Sao Paulo Governor Joao Doria, a leading Bolsonaro opponent.
The president has thrown his support behind another vaccine, developed by Oxford University in Britain and the British-Swedish pharmaceutical firm AstraZeneca.

02:23
Brazilian state of Sao Paulo builds plant to make China’s coronavirus vaccine candidate, CoronaVac
Even as it reversed course, Anvisa stood by its decision, which it said was purely “technical,” in a statement mainly devoted to defending the agency against criticism of political interference in the medical regulatory process.
It said the public health centre coordinating the study in Brazil, the Butantan Institute, had not sent it the cause of death, independent safety review or a required incident report until Tuesday.
The decision “took into consideration the data known to the agency at the time,” it said.
Additional reporting by Eduardo Baptista

.png?itok=arIb17P0)