Coronavirus: WHO backs AstraZeneca vaccine after South Africa delays jabs
- The AstraZeneca shot has run into several setbacks, including concerns about its efficacy against a Covid-19 variant, and its suitability for people over 65
- The vaccine accounts for almost all of the 337.2 million vaccine doses the WHO-led Covax scheme is preparing to begin shipping to some 145 countries
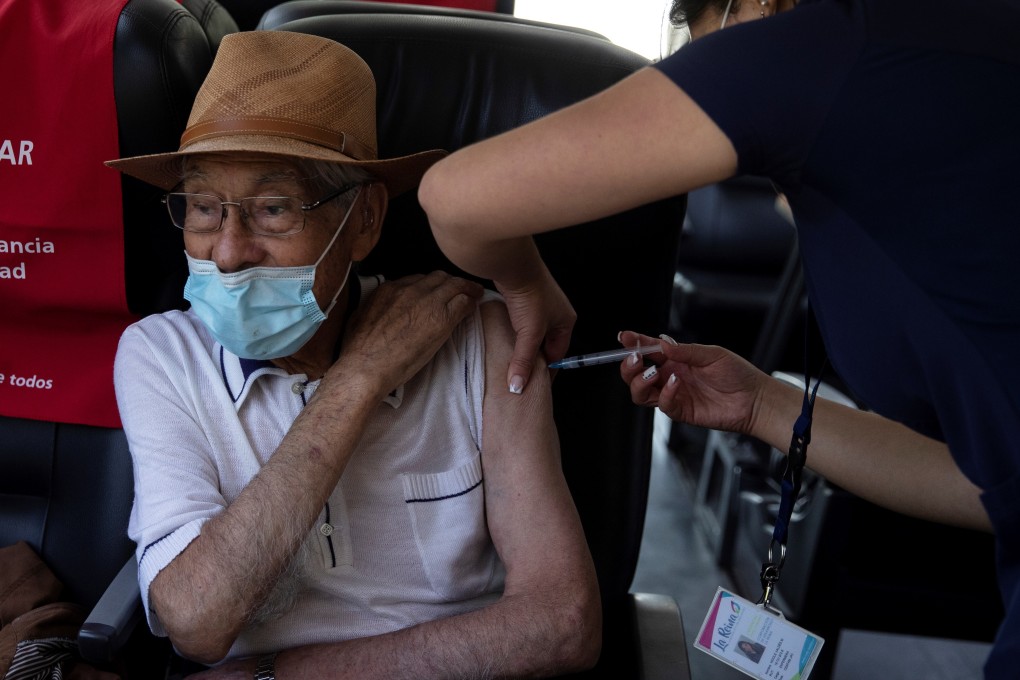
The World Health Organization insisted on Monday that the AstraZeneca vaccine was still a vital tool in the global fight against the coronavirus pandemic, after South Africa delayed the start of its inoculation programme over concerns about its efficacy against a virus variant.
“It is vastly too early to be dismissing this vaccine,” said Richard Hatchett, who heads the Coalition for Epidemic Preparedness Innovations (CEPI), which co-leads the Covax vaccine facility with the WHO and Gavi after the AstraZeneca shot ran into a number of setbacks, including questions about its efficacy for over-65s.
“It is absolutely crucial to use the tools that we have as effectively as we possibly can,” he said, speaking at the WHO’s regular bi-weekly press briefing on the coronavirus pandemic.
The AstraZeneca vaccine is currently a vital part of Covax, which was set up to procure Covid-19 vaccines and ensure their equitable distribution around the world.
09:50
SCMP Explains: What's the difference between the major Covid-19 vaccines?
It accounts for almost all of the 337.2 million vaccine doses Covax is preparing to begin shipping to some 145 countries during the first half of the year, once it receives WHO authorisation, which is expected next week.
But a trial at Johannesburg’s University of Witwatersrand, concluded the vaccine provided only “minimal” protection against mild to moderate Covid-19 caused by the variant first detected in South Africa.