AstraZeneca Covid-19 drug neutralises Omicron sub-variants, says lab study
- This is the first data looking at impact of AstraZeneca’s Evusheld treatment on Omicron variant ‘cousins’ following recent global spike in infections
- Therapy reduced viral load of tested sub-variants in mice lungs; Evusheld contains lab-made antibodies designed to linger in body for months
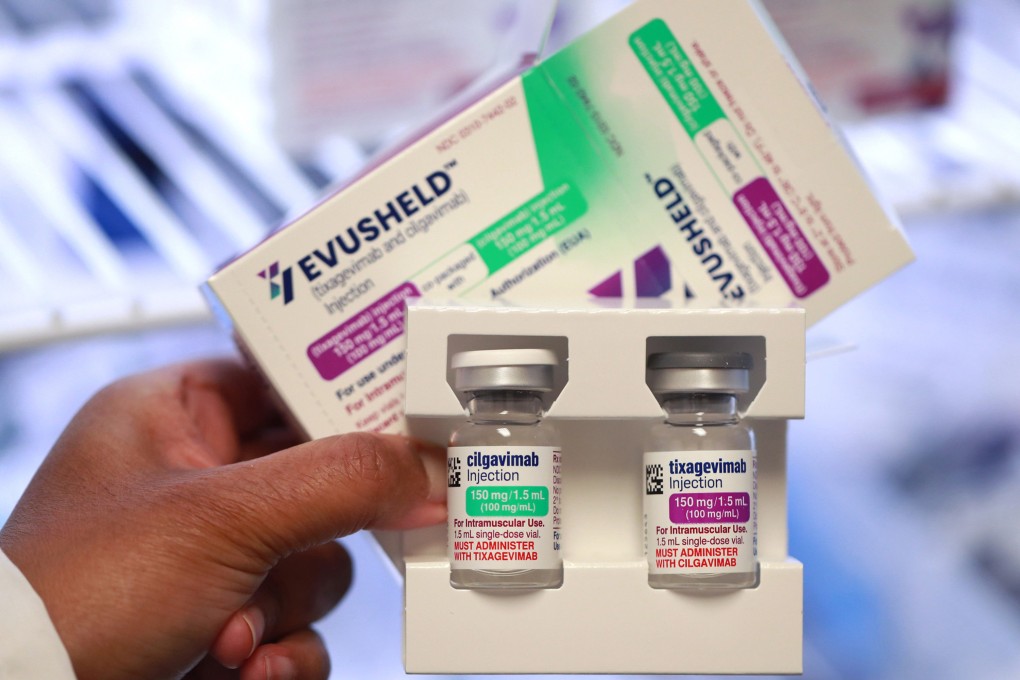
AstraZeneca said on Monday its antibody-based cocktail to prevent and treat Covid-19 retained neutralising activity against Omicron coronavirus variants, including the highly contagious BA. 2 sub-variant, in an independent lab study.
This is the first data looking at the impact of AstraZeneca’s Evusheld treatment on “cousins” of the Omicron variant following a recent global spike in cases. The Anglo-Swedish drug maker said in December that another lab study found that Evusheld retained neutralising activity against Omicron.
Data from the latest study by Washington University in the United States showed the therapy reduced the amount of virus detected in samples – viral load – of all tested Omicron sub-variants in mice lungs, AstraZeneca said. The study has yet to be peer reviewed.
“The findings further support Evusheld as a potential important option to help protect vulnerable patients such as the immunocompromised who could face poor outcomes if they were to become infected with Covid-19,” said John Perez, head of Late Development, Vaccines & Immune Therapies at AstraZeneca.