Advertisement
First human-pig embryos open route to growing transplant organs in chimeras
Researchers ensure that embryos aren’t born with brains
3-MIN READ3-MIN
By Marcus Strom
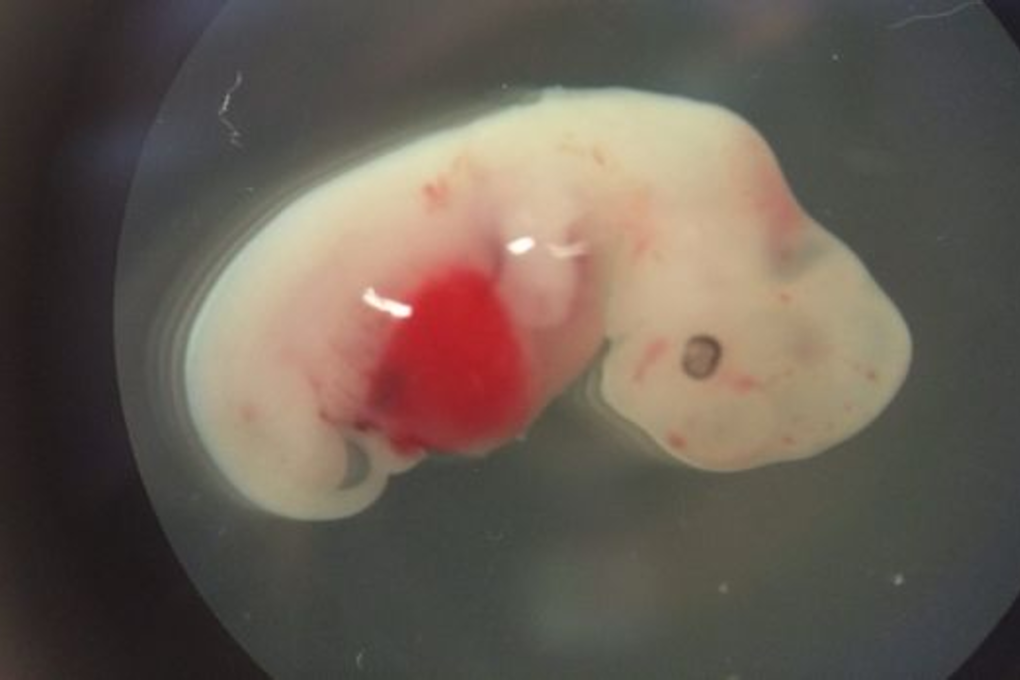
Scientists have grown the first human-pig hybrid embryos and taken them a third of the way through pregnancy inside a sow.
Geneticists at the Salk Institute in California hope this research will pave the way to grow fully-functioning human transplant organs inside hybrid animals.
Advertisement
That, however, is a long way off. The immediate use for this research promises to revolutionise drug testing and help create personalised medicine for treatment of cancers and genetic diseases.
“The ultimate goal of this research is to generate organs that can match to your immune system,” said Dr Jun Wu from the Salk Institute, lead author of the research.
Advertisement
Advertisement
Select Voice
Select Speed
1.00x