Advertisement
OpinionIndonesia’s Sinovac coronavirus vaccine trial raises safety, affordability concerns
- President Joko Widodo’s demand for a vaccine to be available within months has sparked fears the country is rushing ahead without proper precautions
- There are also concerns over cost and availability should the trials prove successful and whether ordinary Indonesians are being used as ‘human guinea pigs’
3-MIN READ3-MIN
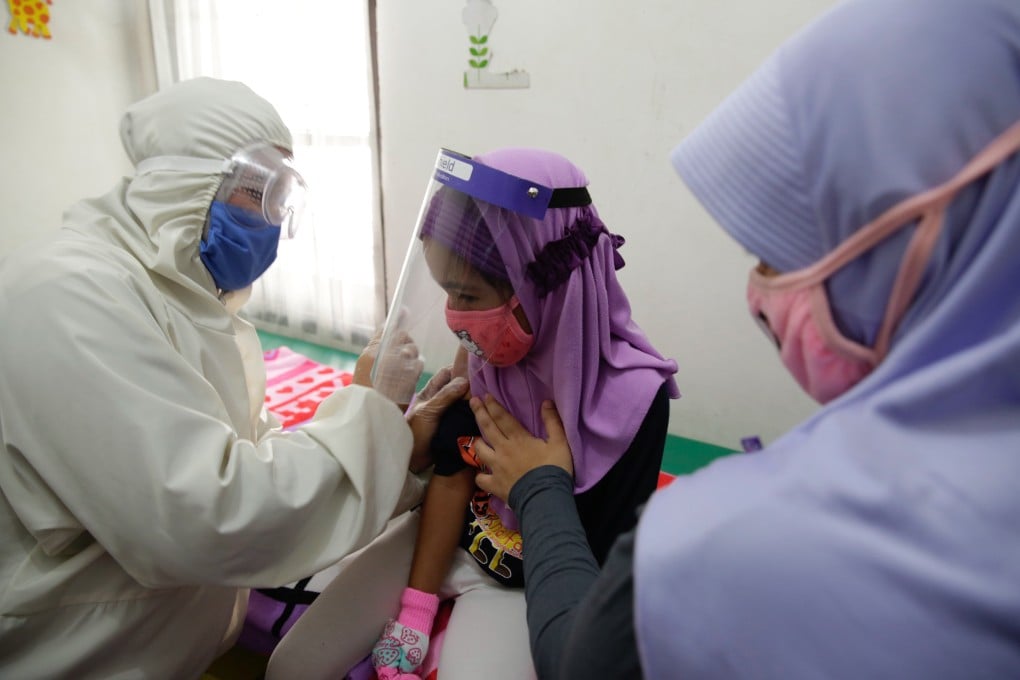
Concerns are growing in Indonesia over a coronavirus vaccine being rushed to development in partnership with China’s Sinovac Biotech, and whether everyone in the country will have access to it even if it does prove effective.
The vaccine candidate known as CoronaVac is set to be tested on some 1,620 volunteers in Indonesia starting this week, making it among only a handful in the world to enter large-scale clinical trials on humans. Sinovac has either started or plans to carry out similar trials in Brazil, Turkey, Bangladesh and Chile, with the firm saying it needs volunteers from outside China as the pandemic has largely been contained within the country’s borders.
Indonesia, the world’s fourth most populous country, has been struggling to contain its mounting virus cases, with more than 127,000 confirmed infections and over 5,700 deaths so far.
Advertisement
President Joko Widodo said in July that he wanted a Covid-19 vaccine to be available within three months, sparking fears that the government is rushing ahead with a process that would normally take years in a bid to fully reopen the country’s economy.

Advertisement
Cabinet Secretary Pramono Anung has said Indonesia hopes to begin mass production of the vaccine by the turn of the year, although health officials in other countries have warned that such a timetable is likely impossible.
Advertisement
Select Voice
Select Speed
1.00x