How parasitic worms could be used against multiple sclerosis and other serious diseases
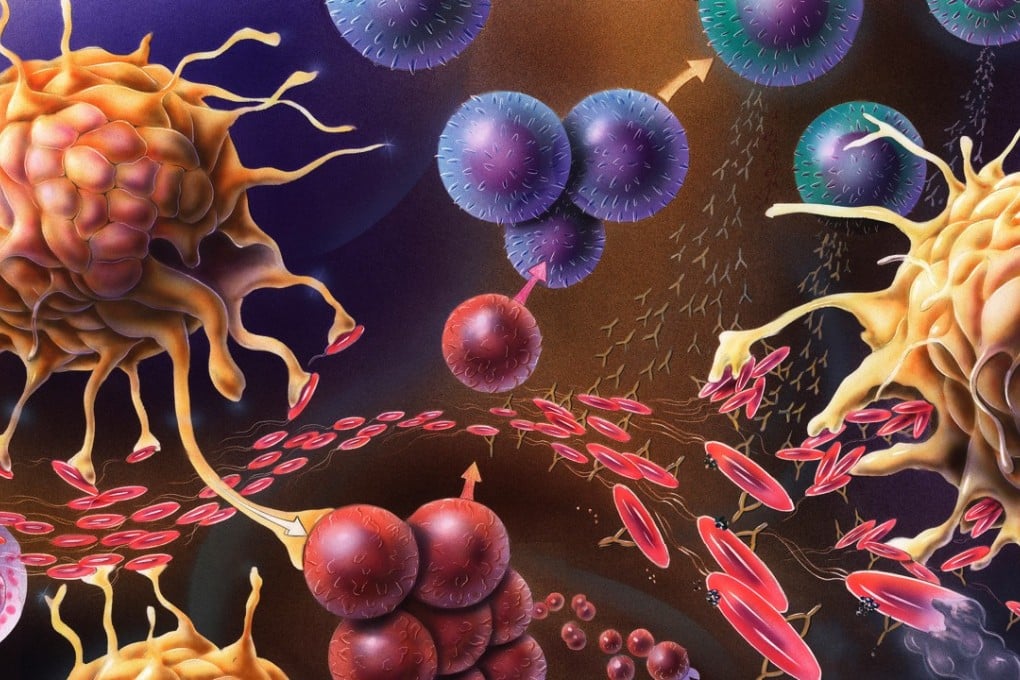
No one wants a parasitic worm infection and they can be bad for your health, but studies show diseases such as asthma, multiple sclerosis and Crohn’s disease can be reduced if your body is a host to these blood drinkers
By Jamie Morton
It’s a disgusting but true fact that humans have long lived with parasites that dwell within our bodies, feeding off us and sometimes causing sickness and disease.
Today, it’s estimated a billion people are infected by parasitic worm infections, mostly in developing nations.
Tapeworm in Chinese man’s brain four years removed by British scientists
Hookworms routinely infect humans through the skin – usually the foot – before migrating to the lungs and eventually establishing themselves in the gut where they can persist for years, feeding on blood. That can lead to iron and blood deficiency in the human host, affecting growth and development, as well as fatigue and general weakness.
But it wasn’t always just poorer countries affected by them – and it was only relatively recently that Western populations shed the worms they’d long carried.
“We were evolved to carry parasites all of the time – from birth to death,” explains Professor Graham Le Gros, a world-leading immunologist and the director of Wellington’s Malaghan Institute of Medical Research. “The simple introduction of sanitation and drugs that cleared the worms from our intestine stopped the reinfection cycle, and we essentially became worm free overnight.”
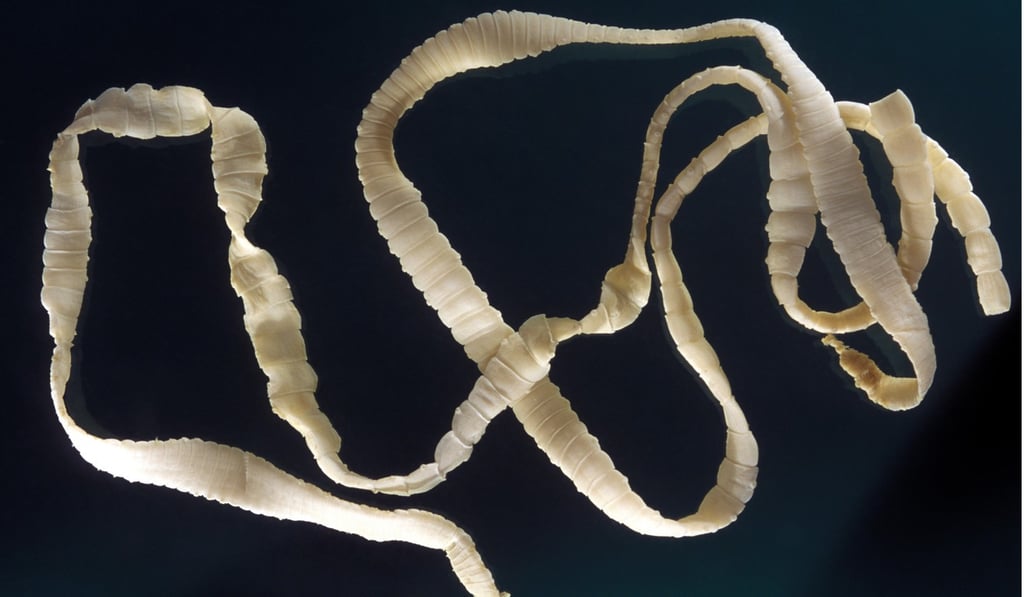
Some of these parasites are much more ghastly than others. The tapeworm can grow up to 15 metres long and live inside a human for three decades. But the hookworm, measuring just a few millimetres and estimated to affect 500 million people, is barely noticeable if in small enough numbers.