75,000 doses of Sanofi Pasteur flu vaccine from batch containing impurities have been used in Hong Kong
- All flu vaccination services suspended at public hospitals and outpatient clinics, authorities say
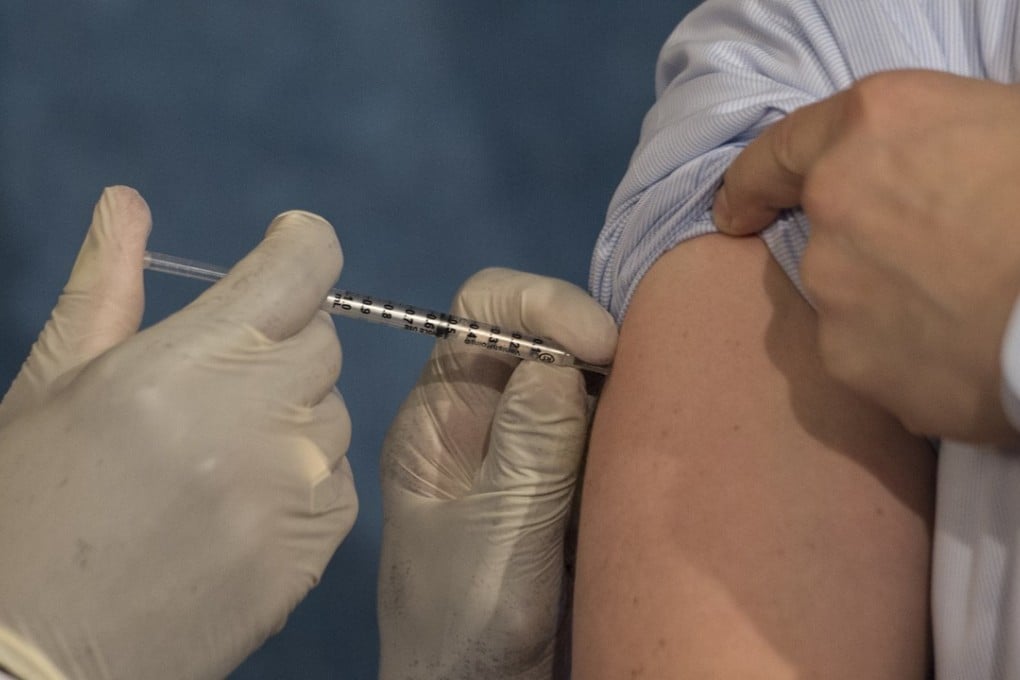
About 75,000 doses of imported flu vaccine belonging to a batch containing impurities have been administered in Hong Kong, the city’s health authorities disclosed on Tuesday.
The Department of Health said 175,000 doses from a batch containing “white particles” had been delivered to the city’s public health care providers from French manufacturer Sanofi Pasteur.
All flu vaccination services had therefore been suspended at hospitals and outpatient clinics, but were expected to resume gradually from Saturday, the Hospital Authority said.
“There has been no adverse reaction reported related to vaccinations with the affected batch,” the authority added. “Members of the public feeling unwell after vaccination should seek medical advice.
“The authority will continue to liaise closely with the Department of Health for an update on the latest situation.”